Eating for Energy: The Science of Tricaprin and GLP-1
Your body already makes the world's most effective energy-regulating hormone. C10 Tricaprin tells it when to release — triggering a cascade of clean, sustained fuel that drug-based GLP-1 therapies try to replicate artificially.
You've probably heard of GLP-1 — the hormone behind Ozempic and Wegovy. What most people don't realize is that your body produces GLP-1 naturally, every single day. And what you eat determines whether that natural supply gets switched on or quietly switched off.
This is the story of how C10 — the fatty acid at the heart of Tricaprin — works not as a drug but as a dietary signal: a natural trigger that tells your gut to release GLP-1, which in turn helps stabilize blood sugar, regulate appetite, protect your heart, and deliver the kind of clean, sustained energy that most people over 50 haven't felt in years.
Two landmark studies published in Nature's family of journals in 2025 brought Tricaprin to global attention — but the GLP-1 pathway was already being mapped by Japanese researchers years earlier. In this article, we connect those dots clearly and honestly, including where the research is solid and where it remains preliminary.
What Is GLP-1 — and Why Does It Matter for Energy?
GLP-1 (Glucagon-Like Peptide-1) is a hormone produced by specialized cells in your small intestine called L-cells. Its release is triggered by food — specifically, by certain nutrients making contact with receptors in your gut wall. Once released, GLP-1 travels through your bloodstream and acts on multiple organs simultaneously.
For the purposes of energy and muscle metabolism, GLP-1 does three things that matter enormously as we age:
GLP-1 signals the pancreas to release insulin only when glucose is actually present — preventing the spikes and crashes that drain energy and damage muscle tissue over time
GLP-1 receptors in the heart and blood vessels reduce inflammation and support healthy circulation — the same mechanism that made GLP-1 drugs compelling in cardiovascular trials
GLP-1 crosses the blood-brain barrier and activates satiety centers — reducing hunger without the jitteriness of stimulants, creating a calmer, more stable energy state
The challenge is that GLP-1 has an extremely short life in the body — naturally degraded by an enzyme called DPP-4 within about two minutes of release. Pharmaceutical GLP-1 drugs work by mimicking this hormone with a longer-lasting synthetic version. But the body's own GLP-1 system is still active — it just needs the right dietary trigger to fire.
Endogenous GLP-1 (the kind your body makes naturally) acts locally — in the gut wall and portal vein — before it ever reaches systemic circulation. Even small, short-lived increases in gut GLP-1 have been shown to produce significant effects on glucose tolerance and appetite. This is why the right food signal can matter as much as a drug — it triggers GLP-1 exactly where and when it's needed most.
Why Does Natural GLP-1 Production Decline With Age?
As we age, L-cell sensitivity to food signals declines. The gut becomes less responsive to nutrients, secreting less GLP-1 after meals than it did decades earlier. The result is a dampened metabolic signal: blood sugar rises higher after eating, stays elevated longer, and the calming, energy-stabilizing effect of natural GLP-1 weakens. For people over 50, this contributes directly to the classic post-meal energy crash, creeping weight gain around the midsection, and the gradual loss of muscle mass associated with sarcopenia.
This is where C10 becomes relevant — not as a drug, but as a precise dietary signal that reactivates the gut's own GLP-1 machinery.
The GPR84 Pathway: How C10 Triggers Your Gut to Release GLP-1
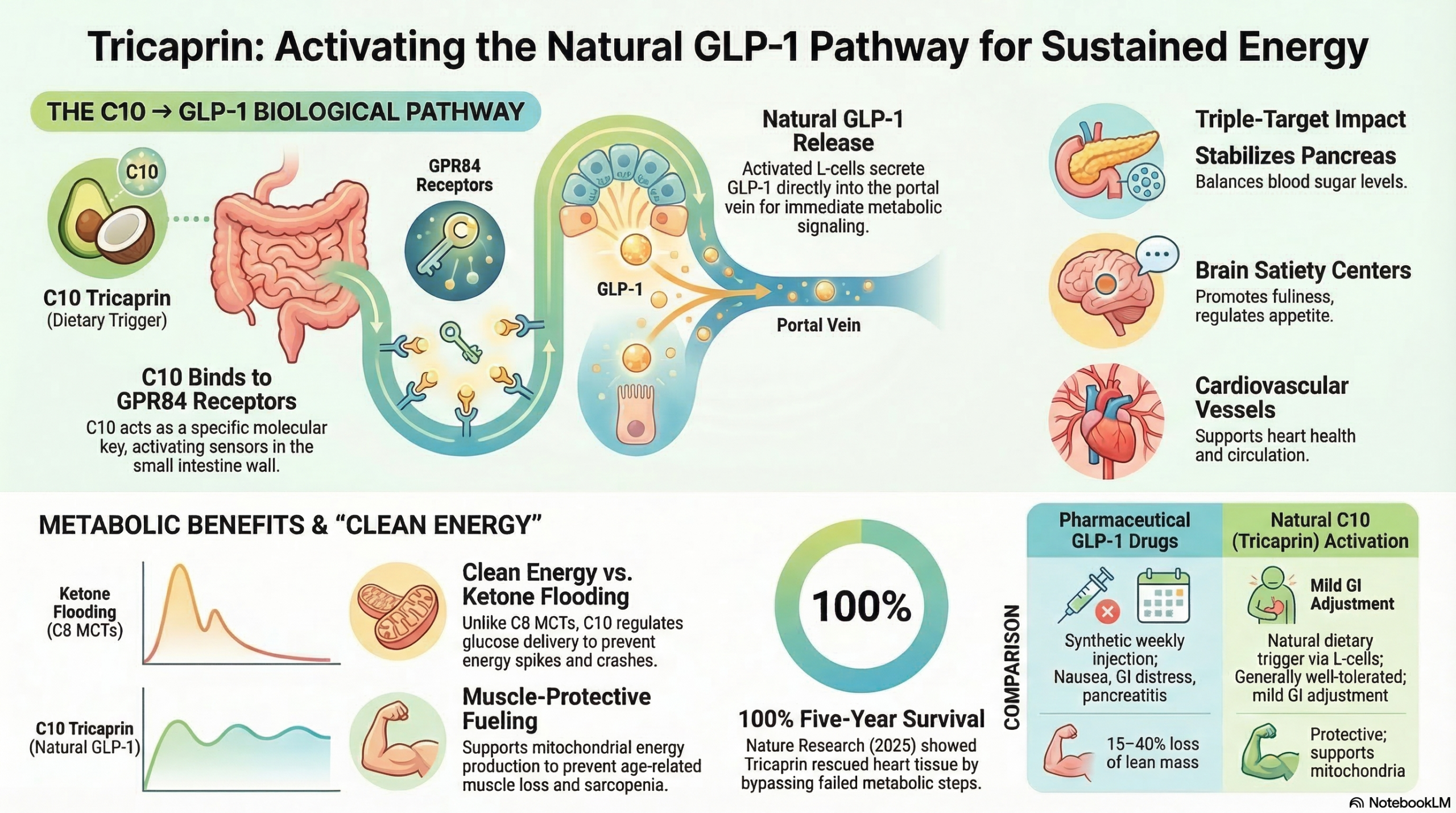
The discovery of the specific mechanism by which C10 stimulates GLP-1 release is one of the most elegant pieces of nutrition science of the past decade. It involves a receptor most people have never heard of — GPR84.
GPR84 is a G-protein coupled receptor embedded in the wall of your intestine. It is essentially a molecular lock that detects the presence of medium-chain fatty acids passing through the gut. When C10 (decanoic acid) binds to GPR84, it acts as a key — triggering a signaling cascade inside the L-cell that results in the secretion of GLP-1 into the bloodstream and portal vein.
Critically, this is a C10-specific effect. The 2022 Frontiers in Nutrition study by Nonaka et al. tested C8, C10, and C12 individually, and found that only C10 produced this potent GPR84-mediated GLP-1 response. C8 did not activate GPR84 in the same way. This is why Tricaprin — as a pure C10 triglyceride — is uniquely positioned to deliver this gut hormone effect, and why standard MCT oil blends (which are mostly C8) cannot reliably replicate it.
C10 Is Consumed & Reaches the Small Intestine
Unlike long-chain fats that require bile salts and travel through the lymphatic system, C10 is absorbed directly through the gut wall and transported via the portal vein — arriving rapidly at the site of action.
C10 Binds to GPR84 Receptors on L-Cells
Intestinal enteroendocrine L-cells express GPR84 — a receptor specifically tuned to detect medium-chain fatty acids. C10 binds to GPR84 as a potent agonist, activating the receptor's intracellular signaling cascade.
L-Cells Release GLP-1 Into the Portal Circulation
Activated L-cells secrete GLP-1 directly into the gut and portal vein. Even before GLP-1 reaches systemic circulation, this local portal GLP-1 signal produces strong effects on glucose uptake and insulin sensitivity in the liver and pancreas.
GLP-1 Signals Three Organs Simultaneously
Pancreas: releases insulin proportionally to blood glucose — no overshot, no crash. Brain: activates satiety signals, reducing appetite and food-seeking behavior. Heart & vessels: reduces inflammation, improves circulation.
Result: Stable Blood Sugar + Sustained Clean Energy
Glucose enters cells steadily and efficiently. Mitochondria receive consistent fuel. The energy "crash" from blood sugar spikes is avoided. Muscle tissue is protected from catabolic breakdown during low-glucose episodes.
The GPR84 → GLP-1 mechanism described above has been demonstrated in mouse models and mouse enteroendocrine cell lines (Nonaka et al., 2022; Scientific Reports, 2025). The specific pathway is mechanistically well-characterized, but large-scale human clinical trials confirming this exact mechanism in humans have not yet been published. We present this research accurately and believe the findings are compelling — but we distinguish clearly between animal/cell-line evidence and confirmed human outcomes.
What the 2025 Nature Studies Tell Us About "Clean Energy"
The phrase "clean energy" gets used loosely in wellness marketing. In the context of Tricaprin and GLP-1, it has a specific, scientifically grounded meaning — and two landmark 2025 publications help illustrate why.
Scientific Reports (2025): TC10, GLP-1, and Cognitive Protection
TC10 Attenuates Cognitive Decline Via the GLP-1 Pathway
Researchers administered TC8 and TC10 to mice on a high-fat diet and measured cognitive outcomes and metabolic markers. TC10 improved glucose tolerance — but crucially, this effect was GLP-1-dependent. When GLP-1 signaling was blocked, TC10's cognitive protective effects were significantly attenuated. This was described as the first study to specifically link TC10's brain-protective effects to the GLP-1 pathway — rather than to ketone production (TC8's mechanism). The study also confirmed TC10 reduced blood glucose levels in glucose tolerance testing.
Read the full paper → Scientific Reports, March 2025This is where the "clean energy" concept becomes tangible. TC8 (like standard MCT oil) produces energy by flooding the liver with ketone bodies — a rapid but somewhat blunt fuel signal that can produce spikes and, for some people, GI discomfort. TC10's GLP-1 pathway does something more precise: it regulates the delivery of glucose to cells rather than bypassing glucose entirely. The result is energy that is stable, smooth, and sustained — not the caffeine-like rush and crash of ketone flooding.
Nature Cardiovascular Research (2025): Energy Failure at the Heart of Disease
100% 3-Year Survival in TGCV Patients Treated With Tricaprin
The Hirano et al. 2025 study in Nature Cardiovascular Research documented remarkable outcomes in patients with Triglyceride Deposit Cardiomyovasculopathy (TGCV) — a condition where defective intracellular fat breakdown causes cellular energy failure in heart muscle. Tricaprin-treated patients achieved 100% 3-year survival vs. 78.6% in controls — and 100% 5-year survival vs. 68.1%. The mechanism: Tricaprin corrects the cellular energy deficit by replacing long-chain fat (which these patients cannot process) with C10 that bypasses the broken metabolic step entirely.
Read the full paper → Nature Cardiovascular Research, 2025The relevance for healthy aging adults is this: TGCV is an extreme version of the same cellular energy failure dynamic that occurs gradually in aging muscle cells. The mechanism — blocked fat metabolism leading to mitochondrial fuel starvation — is the same. Tricaprin's correction of that failure in cardiac tissue demonstrates that the bypass mechanism works under the most demanding conditions imaginable. In muscle cells facing the milder but cumulative energy deficits of aging, the same logic applies.
In the context of these two 2025 studies, "clean energy" has a precise definition: energy delivered to cells through a metabolic pathway that does not generate the inflammatory byproducts, oxidative stress, or blood sugar instability associated with poor-quality fuel. C10 delivers clean energy by bypassing blocked metabolic steps, triggering GLP-1 to optimize glucose handling, and activating mitochondrial machinery to burn that fuel efficiently — all without stimulants, without blood sugar spikes, and without the catabolic stress that ages muscle tissue prematurely.
Natural C10 GLP-1 vs. Pharmaceutical GLP-1 Drugs: What's the Difference?
It's important to be honest here: pharmaceutical GLP-1 drugs (Ozempic, Wegovy, Mounjaro) are powerful, clinically validated medications for obesity and type 2 diabetes. C10 Tricaprin is a dietary supplement that stimulates your body's own GLP-1 production. These are not the same thing — and for people with serious metabolic disease, medication is often the right choice. But for adults over 50 focused on energy, muscle preservation, and long-term metabolic health, the comparison is worth understanding.
| Feature | Drug GLP-1 (Ozempic/Wegovy) | Natural C10 via Tricaprin |
|---|---|---|
| GLP-1 activation type | Synthetic GLP-1 analogue injected weekly | Stimulates endogenous GLP-1 from L-cells via food signal |
| Duration of effect | ~1 week (long-acting agonist) | Tied to meals — activates with each C10-containing dose |
| Muscle mass effect | ⚠ Risk — 15–40% of weight lost is lean mass | ✓ Protective — C10 supports mitochondrial fuel and muscle preservation |
| Blood sugar management | ✓ Strong — clinically validated for T2DM | ~ Moderate — animal evidence; human glucose data emerging |
| Side effects | Nausea, vomiting, GI distress, pancreatitis risk | Mild GI adjustment at high doses; generally well tolerated |
| Mitochondrial effect | No direct mitochondrial mechanism | ✓ Direct — PPARγ, Complex I, SIRT1 activation (see Article 2) |
| Requires prescription? | Yes | No — available as dietary supplement |
| Best suited for | Obesity (BMI 30+), type 2 diabetes | Adults 50+ focused on energy, muscle health, metabolic support |
The bottom line: these tools serve different populations with different needs. What is notable is that where pharmaceutical GLP-1 drugs carry significant risk for muscle loss — one of the most serious concerns for adults already facing sarcopenia — C10's combination of GLP-1 stimulation and direct mitochondrial support works in the opposite direction. It is metabolically complementary rather than metabolically depleting.
The Research Behind These Claims
Five peer-reviewed studies form the evidentiary foundation of this article.
C10 Stimulates GLP-1 via GPR84 — The Foundational Mechanism
Nonaka et al. (2022) in Frontiers in Nutrition demonstrated that C10 (but not C8) stimulates GLP-1 secretion via GPR84 activation in mouse enteroendocrine cells and in vivo. C10 improved glucose tolerance, reduced HFD-induced obesity, and increased plasma GLP-1 and insulin levels. Effects were attenuated in GPR84-deficient mice, confirming the receptor dependency. This is a mouse/cell-line study.
Read the Study → Frontiers in Nutrition, 2022TC10 Attenuates Cognitive Decline via GLP-1 (2025)
This 2025 Scientific Reports (Nature Publishing Group) study is the first to demonstrate that TC10's cognitive protective effects in mice are GLP-1-dependent — distinguishing TC10's mechanism from TC8's ketone-based pathway. TC10 also reduced blood glucose in glucose tolerance testing. The GLP-1 connection to cognitive and metabolic protection was directly confirmed. This is a mouse study.
Read the Study → Scientific Reports, March 2025Tricaprin Delivers Clean Cellular Energy Where Normal Metabolism Fails
Hirano et al. (2025) in Nature Cardiovascular Research documented 100% 3-year and 5-year survival in tricaprin-treated TGCV heart failure patients — compared to 78.6% and 68.1% in controls. The underlying mechanism is cellular energy rescue: Tricaprin replaces the failed long-chain fat metabolism pathway with C10's direct mitochondrial entry. This is the strongest human-outcome evidence for Tricaprin's metabolic rescue capability.
Read the Study → Nature Cardiovascular Research, 2025Human MCT Trial: Real-World Energy and Muscle Outcomes
Ezaki & Abe (2023) in Frontiers in Nutrition — a combined analysis of 3 clinical trials — found that 6g/day of C8/C10 MCTs over 3 months improved grip strength, walking speed, and muscle mass in frail older adults (mean age 85). These outcomes reflect the integrated energy and muscle-preservation effects that the GPR84/GLP-1 and mitochondrial mechanisms predict.
Read the Study → Frontiers in Nutrition, 2023GLP-1: The Glucose Homeostasis Master Regulator
The foundational understanding of how endogenous GLP-1 regulates blood glucose — slowing gastric emptying, enhancing glucose-dependent insulin secretion, suppressing glucagon — comes from decades of incretin research. Notably, even small increases in portal GLP-1 have disproportionately large effects on glucose tolerance, making the gut-specific C10 → GPR84 → L-cell mechanism highly relevant even without systemic GLP-1 elevation.
GLP-1 Regulation Review → PMC, 2014Practical Guide: Eating for GLP-1 with C10 Tricaprin
If your goal is steady, clean energy — the kind that doesn't crash after lunch and doesn't require caffeine to sustain — the following protocol reflects what the research suggests is most likely to activate the GLP-1 pathway with C10.
Take C10 With Your First Real Meal of the Day
The GPR84 receptor in your gut responds to C10 arriving with food — not on an empty stomach. Taking C10 Tricaprin with breakfast, mixed into coffee or drizzled onto eggs, avocado, or yogurt positions it to encounter intestinal L-cells while they are primed to respond to nutrients. This is the most likely context for meaningful GLP-1 stimulation.
Include Fiber and Protein in the Same Meal
GLP-1 release is synergistically triggered by dietary fiber and protein — both of which independently activate L-cell receptors. A breakfast that combines C10, high-fiber vegetables or oats, and quality protein (eggs, Greek yogurt, or legumes) creates multiple GLP-1 signals arriving together, potentially amplifying the total gut hormone response.
Aim for 5–10g of C10 Per Day — Don't Overdo It
The human muscle trial (Ezaki & Abe, 2023) used just 6g/day — roughly 1–1.5 teaspoons. The GPR84 pathway is a receptor signal, not a dose-volume relationship. More C10 does not mean proportionally more GLP-1. Start at 1 teaspoon, build to 1.5 over 2–4 weeks, and stay consistent rather than escalating dose.
Avoid High-Sugar Meals Immediately After C10
The whole point of the GLP-1 pathway is to deliver glucose to cells gradually and efficiently. If you take C10 and then eat a sugary cereal or pastry, you flood the system before the GLP-1 signal can moderate the response. Pair C10 with lower-glycemic foods to let the pathway do its job.
Track Your Energy at 2 and 4 Hours Post-Meal
The classic marker of better glucose metabolism is the absence of the afternoon energy crash. Most people who adopt this protocol report that their 2–4 pm energy dip becomes less severe within the first 2–3 weeks. Use that as your baseline signal — not weight on the scale, which moves slowly and is affected by many variables.
If You Are on GLP-1 Medication, Speak to Your Doctor First
If you are taking semaglutide, tirzepatide, or another GLP-1 receptor agonist, do not add C10 Tricaprin without medical supervision. While these are different mechanisms, combining dietary GLP-1 stimulation with pharmaceutical GLP-1 agonism warrants clinical oversight — particularly regarding blood sugar and appetite effects.
-
1
C10 → GPR84 → GLP-1 Mechanism — Nonaka H, Ohue-Kitano R, Masujima Y, Igarashi M, Kimura I. "Dietary Medium-Chain Triglyceride Decanoate Affects Glucose Homeostasis Through GPR84-Mediated GLP-1 Secretion in Mice." Frontiers in Nutrition. 2022;9:848450.
DOI: https://doi.org/10.3389/fnut.2022.848450 -
2
TC10, GLP-1 & Cognitive Protection (2025) — "Medium-chain triglycerides tricaprin TC10 and tricaprylin TC8 attenuated HFD-induced cognitive decline in a manner dependent on or independent of GLP-1." Scientific Reports (Nature Publishing Group). 2025.
DOI: https://doi.org/10.1038/s41598-025-94129-4 -
3
Tricaprin & Heart Failure Survival (2025) — Hirano K, et al. "Long-term survival and durable recovery of heart failure in patients with triglyceride deposit cardiomyovasculopathy treated with tricaprin." Nature Cardiovascular Research. 2025;4(3):266–274.
DOI: https://doi.org/10.1038/s44161-025-00611-7 -
4
Human MCT Muscle & Energy Trial — Ezaki O, Abe S. "Medium-chain triglycerides (8:0 and 10:0) increase muscle mass and function in frail older adults: a combined data analysis of clinical trials." Frontiers in Nutrition. 2023;10:1284497.
DOI: https://doi.org/10.3389/fnut.2023.1284497 -
5
GLP-1 & Glucose Homeostasis Review — "Regulation of Glucose Homeostasis by GLP-1." Progress in Molecular Biology and Translational Science. PMC4159612. 2014.
URL: https://pmc.ncbi.nlm.nih.gov/articles/PMC4159612/

