What GLP-1 Drugs Don't Tell You About Muscle Loss — And What C10 Does Differently
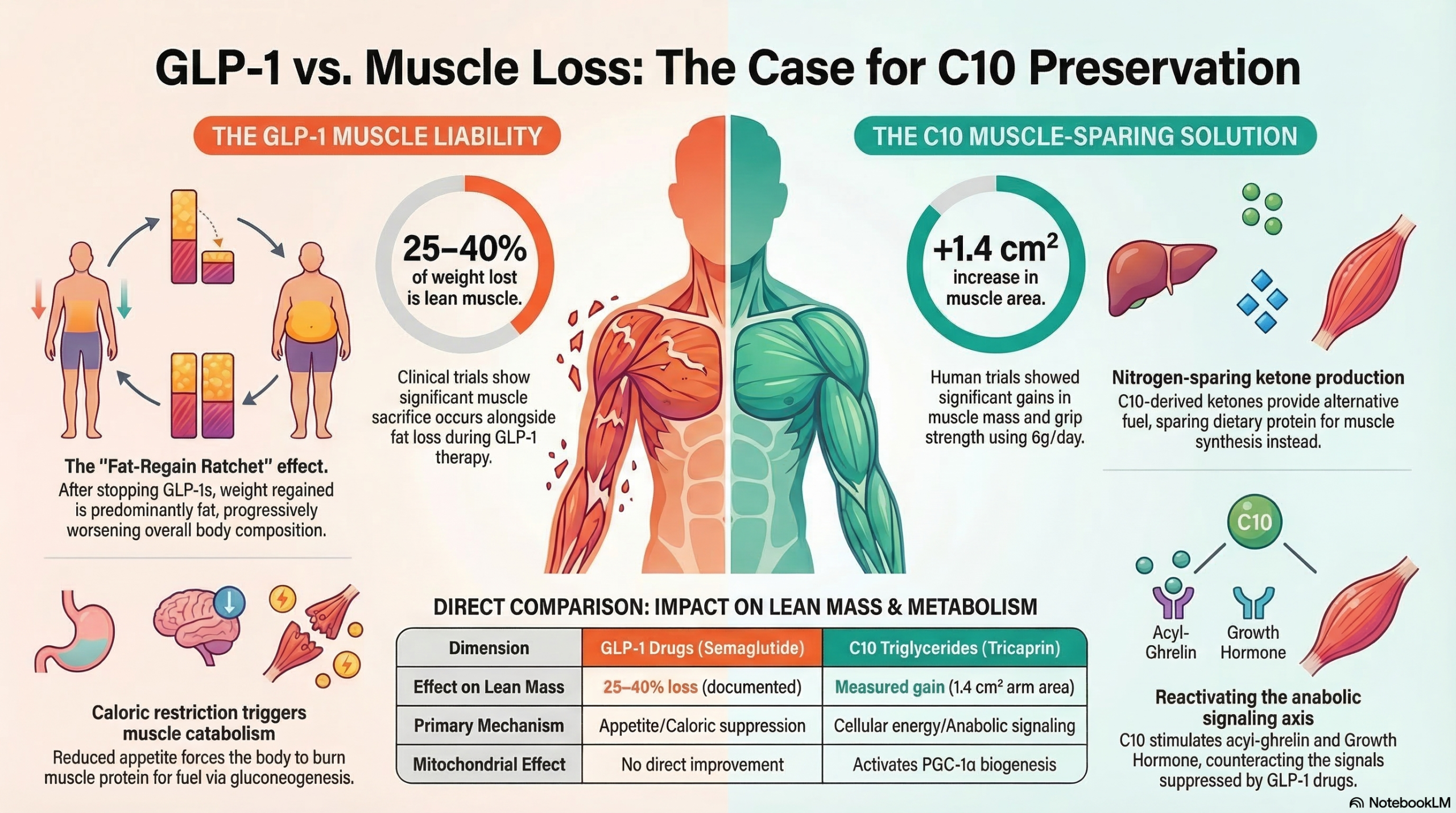
GLP-1 receptor agonists produce dramatic weight loss — but up to 40% of that lost weight is muscle, not fat. The mechanism is predictable and well-documented. So is the solution. Here is what the clinical data shows, and why C10 and GLP-1 drugs address different problems rather than competing ones.
GLP-1 receptor agonists — semaglutide (Ozempic, Wegovy), tirzepatide (Mounjaro, Zepbound), and the drugs that will follow — represent the most significant advance in obesity pharmacology in a generation. The clinical trial data on weight loss and cardiometabolic outcomes is, in several respects, extraordinary. The cardiovascular outcome trials have exceeded most expectations.
None of that is in dispute here.
What is less prominently discussed — in prescriber conversations, in consumer marketing, and in much of the popular health media — is that a meaningful fraction of the weight lost on GLP-1 drugs is lean muscle mass. Not fat. Muscle. And the cellular mechanism driving that muscle loss is not a side effect that will be engineered away in the next formulation. It is an inherent consequence of how caloric restriction works on muscle tissue when protein intake drops and anabolic signaling weakens.
This article examines what the clinical evidence says about GLP-1-related lean mass loss, why it happens, what the downstream consequences are, and what C10 triglycerides do — mechanistically and in clinical trials — that addresses the problem from the cellular level up.
This is not an argument against GLP-1 drugs. The cardiovascular and metabolic outcome data for semaglutide and tirzepatide is substantial and in many patients represents a clinically important benefit. This article examines a specific, documented limitation of GLP-1 pharmacotherapy — lean mass loss — and presents the mechanistic and clinical case for C10 as a complementary intervention. The two are not in competition.
The Lean Mass Loss Problem — What the Trials Actually Show
The STEP and SURMOUNT trial series — the registration-quality Phase III trials for semaglutide and tirzepatide — produced headline weight loss numbers that drove the GLP-1 boom: 15–21% body weight reduction over 68–72 weeks. These are real numbers from well-conducted randomized trials. They deserve their prominence.
What receives less prominent display is the body composition data embedded in those same trials. When researchers separated total weight loss into fat mass and lean mass components, the picture became more complicated.
To put this in clinical context: a person who loses 15 kg on semaglutide over 68 weeks may lose 4–6 kg of that as lean mass. At the same time, natural sarcopenic muscle decline in adults over 60 runs at roughly 1–2% of total muscle mass per year. A GLP-1 course that removes 4–6 kg of lean mass in 16 months represents a sarcopenic acceleration that could take several years to recoup — if it is recouped at all in an older adult without aggressive countermeasures.
was lean mass
It is worth noting that a 39% lean mass fraction is not dramatically different from what is observed with any significant caloric restriction without specific muscle-preservation strategies. The issue is not unique to GLP-1 drugs — it is inherent to aggressive caloric restriction. GLP-1 drugs are notable because they produce a degree of restriction that most people cannot sustain through lifestyle alone, making the lean mass problem more clinically consequential at scale.
GLP-1 drugs require continuous administration to maintain weight loss. Clinical trial data consistently shows that weight regain begins when the drug is discontinued, and that the regained weight is predominantly fat — not the muscle that was lost. This ratchet mechanism — losing mixed lean and fat on the drug, regaining predominantly fat after discontinuation — means that serial GLP-1 cycles can progressively worsen body composition even as scale weight returns to baseline.
Why GLP-1 Drugs Cause Muscle Loss — The Mechanism
GLP-1 drugs do not directly catabolize muscle. They suppress appetite, reduce caloric intake, and slow gastric emptying. The muscle loss is an indirect consequence — but it is mechanistically predictable from first principles of muscle biology under caloric restriction.
Protein Intake Falls With Appetite Suppression
Skeletal muscle protein turnover is in constant equilibrium between synthesis (MPS) and breakdown (MPB). The net balance — anabolism or catabolism — is driven primarily by protein availability and anabolic signaling. When appetite is suppressed by 30–40%, total caloric and protein intake drops proportionally. Without deliberate, effortful protein intake targeting, protein consumption on GLP-1 drugs is often inadequate to maintain positive muscle protein balance — particularly in older adults who already have diminished anabolic sensitivity to dietary protein (anabolic resistance).
Caloric Deficit Activates Protein Catabolism for Gluconeogenesis
In a sustained caloric deficit, the body increasingly turns to amino acids — drawn from muscle protein — for gluconeogenesis to maintain blood glucose. This is a conservation mechanism: the liver preferentially synthesizes glucose from available amino acids when dietary carbohydrate and fat intake drops. GLP-1-driven caloric restriction, even when accompanied by adequate dietary protein, still creates the substrate conditions that favor muscle catabolism as a glucose source — particularly in the overnight fasted state.
Anabolic Hormone Signals Weaken With Caloric Restriction
Growth hormone, IGF-1, and insulin — the primary anabolic hormones that drive muscle protein synthesis — all decline with aggressive caloric restriction. Insulin, in particular, is acutely anti-catabolic in muscle tissue: it suppresses MPB by inhibiting the ubiquitin-proteasome pathway. In a GLP-1-driven hypocaloric state, reduced postprandial insulin levels diminish this anti-catabolic protection. The net effect is that the anabolic machinery is running at reduced capacity precisely when it needs to be protecting lean mass.
Mitochondrial Demand Signals Are Absent Without Resistance Exercise
Muscle is a metabolically expensive tissue that the body maintains only when signaled to do so. The primary signals for muscle retention are mechanical loading (resistance exercise) and energy demand. GLP-1 drugs reduce appetite but do not improve exercise capacity or motivation. Most patients on GLP-1 drugs who are not already in a structured exercise program do not significantly increase resistance training during their course — which means the "justify this muscle tissue" signal is not being sent, and the body is free to sacrifice it in service of the caloric deficit.
Every mechanism above is amplified in adults over 60. Anabolic resistance — the reduced ability of aging muscle to respond to dietary protein and anabolic hormones — means older adults need more protein per kilogram to achieve the same MPS response as younger adults. GH and IGF-1 are already declining. Mitochondrial function is already compromised. A GLP-1-driven caloric restriction in a 65-year-old is a fundamentally different metabolic event than the same restriction in a 35-year-old. The same drug, in the same dose, with the same caloric deficit, produces disproportionately more lean mass loss in the older patient.
What C10 Does Differently — The Mechanism Contrast
C10 triglycerides (tricaprin) do not suppress appetite, do not create a caloric deficit, and do not produce the cascade of muscle-catabolic consequences that follow from caloric restriction. They operate through a completely different set of cellular mechanisms — and those mechanisms target precisely the failures that drive GLP-1-related muscle loss.
Operating Mechanism: Appetite Suppression → Caloric Deficit
- Reduces total protein intake proportionally with caloric suppression
- Creates substrate conditions favoring gluconeogenesis from muscle amino acids
- Reduces postprandial insulin — weakening anti-catabolic muscle protection
- Does not address mitochondrial function in aging muscle
- Does not stimulate GH secretion — anabolic axis remains suppressed
- Does not improve cellular energy production capacity
- Lean mass loss is a predictable, documented consequence
Operating Mechanism: Cellular Energy + Anabolic Signaling
- Nitrogen-sparing: ketone bodies reduce muscle protein catabolism for fuel
- Stimulates acyl-ghrelin → GH secretion, reactivating the anabolic axis
- Activates PGC-1α via SNS adrenergic pathway → mitochondrial biogenesis
- Activates PPARγ → improves cellular fat metabolism efficiency
- Does not create caloric deficit — does not trigger muscle catabolism cascade
- Human RCT evidence: increased lean mass, grip strength, walking speed
- Lean mass gain is a documented outcome in frail elderly trial populations
The contrast is not a matter of degree — it is a matter of direction. GLP-1 drugs, by their primary mechanism, create conditions that favor lean mass loss. C10 triglycerides, by their primary mechanism, create conditions that favor lean mass preservation and growth. The two compounds are operating on different problems in the same patient.
The Nitrogen-Sparing Mechanism — Why C10 Specifically Counters the GLP-1 Deficit
When C10 is metabolized by the liver, it produces ketone bodies — primarily beta-hydroxybutyrate and acetoacetate. Skeletal muscle can oxidize these ketones as a direct fuel source. When muscle cells have an alternative fuel available, they spare dietary protein from being burned for energy — this is the nitrogen-sparing effect. More dietary protein goes to muscle protein synthesis rather than gluconeogenesis.
This is directly relevant to GLP-1 users. In a caloric deficit driven by appetite suppression, amino acids are being drawn from muscle for gluconeogenesis. Adding C10 to the regimen provides an alternative fuel substrate that competes with amino acids for that gluconeogenic role — effectively preserving more protein for muscle maintenance. The intervention addresses the caloric-restriction-driven amino acid drain at the cellular level rather than simply recommending patients "eat more protein" on a drug that actively suppresses their appetite.
GLP-1 drugs suppress ghrelin — the hunger hormone. This is part of their appetite-suppressing mechanism. But ghrelin's acylated form (acyl-ghrelin) also stimulates growth hormone secretion, which drives muscle protein synthesis. GLP-1-mediated ghrelin suppression may therefore compound the lean mass problem by reducing the acyl-ghrelin → GH anabolic signal. C10 has been shown to increase acyl-ghrelin at doses as low as 3g — providing a partial counterbalance to the GH-axis suppression that may accompany GLP-1 therapy.
The Clinical Evidence: C10's Muscle Outcomes in Context
The Ezaki & Abe trial series — three randomized controlled trials conducted at Showa Women's University between 2016 and 2023, with a combined data analysis published in Frontiers in Nutrition — provides the most directly relevant human clinical data for C10 and muscle outcomes. The trial population (frail nursing home residents, mean age 85) is, in one respect, the most challenging population imaginable for muscle intervention: maximum mitochondrial decline, maximum anabolic resistance, minimum physical activity. The results are therefore a conservative lower bound for what C10 does in more capable populations.
90-day trial · Mean age 85
The directional outcome in the Ezaki & Abe data is the opposite of GLP-1-related body composition changes: lean mass up, fat mass down, functional measures improved. This is not a coincidence — it reflects the mechanistic difference between a cellular energy and anabolic signaling intervention (C10) and a caloric restriction intervention (GLP-1 drugs).
Why These Are Not Competing Interventions
The framing of "C10 vs. GLP-1 drugs" is a false dichotomy. They address different problems at different levels of biology. The practical question is not which one to use — it is whether C10 can serve as a muscle-preservation adjunct in people who are already on or considering GLP-1 pharmacotherapy.
| Dimension | GLP-1 Drugs (Semaglutide / Tirzepatide) | C10 Triglycerides (Tricaprin) |
|---|---|---|
| Primary Mechanism | GLP-1R agonism → appetite suppression → caloric deficit | Cellular energy supply + anabolic signaling cascade |
| Effect on Fat Mass | Strong reduction (12–21% body weight) | Modest reduction (trial data: triceps skinfold decrease) |
| Effect on Lean Mass | Loss (25–40% of weight loss is lean) | Gain (+1.4 cm² arm muscle area, grip strength, walking speed) |
| Cardiovascular Outcomes | Major CV outcomes data (SELECT trial: −20% MACE) | Plaque regression (Leren 1966); TGCV survival (Hirano 2025) |
| Anabolic Hormone Axis | Suppresses ghrelin (part of appetite mechanism) | Increases acyl-ghrelin → stimulates GH secretion |
| Mitochondrial Function | No direct effect | PPARγ activation, PGC-1α upregulation, Complex I enhancement |
| Nitrogen-Sparing | Does not provide alternative fuel for gluconeogenesis | Ketone bodies spare muscle protein from gluconeogenesis |
| Practical Administration | Weekly injection or daily oral (varies by drug) | 6g/day mixed into food at a meal — no injection |
| Evidence Quality for Muscle | Body composition sub-studies of Phase III trials | Three small RCTs + combined analysis (n = 56) |
| Drug Interaction Risk | Multiple documented interactions (gastric emptying slows drug absorption) | No documented interactions with GLP-1 drugs |
The case for combination use is mechanistically coherent: GLP-1 addresses the obesity-driven metabolic disease burden through caloric restriction and cardiometabolic pathways; C10 addresses the lean mass loss that is an inherent consequence of that restriction, plus the mitochondrial and anabolic signaling gaps that GLP-1 drugs do not touch. The combination is not additive in a pharmacological sense — it is complementary in a physiological sense.
A Practical Protocol for GLP-1 Users — Preserving Muscle While Losing Weight
The following protocol is built from the Ezaki & Abe trial parameters for C10, current evidence-based recommendations for protein intake during caloric restriction, and resistance exercise guidance for adults on GLP-1 drugs. None of this supersedes guidance from a prescribing physician — it supplements it.
Prioritize Protein at Every Meal — Hit 1.2–1.6g per kg Bodyweight
GLP-1 drugs reduce appetite broadly, and most patients on semaglutide or tirzepatide consume significantly less total protein than they did pre-treatment. Current evidence for muscle preservation during caloric restriction supports a target of 1.2–1.6g of protein per kilogram of bodyweight per day — approximately double the standard RDA. On a drug that makes eating feel difficult, this requires deliberate prioritization: protein-first at every meal, protein-dense snacks when capacity allows, protein shakes if whole food intake is insufficient. This is the single most impactful lifestyle modification for GLP-1 muscle preservation.
🥚 Protein target: 1.2–1.6g/kg/day — protein first at every mealAdd 6g of C10 Daily — With Your Largest Meal
The Ezaki & Abe trial protocol administered 6g of C8/C10 MCTs mixed into food at dinnertime. Starting at 3g and titrating to 6g over two to three weeks reduces the GI adjustment period common with MCT-naive adults. The key parameters: with food (not on an empty stomach), at a full meal where the caloric substrate context supports the ketogenic and GLP-1 secretion cascade that C10 initiates. For GLP-1 drug users already managing nausea, the food-mixed approach is preferable to capsule-form C10 taken with minimal caloric context.
🍽️ 6g C10 mixed into food at your main meal · Start at 3g week 1–2Resistance Train — Minimum 2× Per Week, Any Format
The single most powerful signal for muscle preservation during caloric restriction is mechanical loading. Resistance exercise triggers AMPK-mediated PGC-1α activation independently of C10's SNS-adrenergic pathway — meaning the two signals are additive, not redundant. The threshold for meaningful benefit is low: two sessions per week of bodyweight movements, resistance bands, or light free weights is sufficient to provide the "maintain this tissue" signal that the body needs during a GLP-1-driven caloric deficit. GLP-1 drugs do not directly impair exercise capacity; most patients can maintain or increase activity levels with some adjustment during the early weeks of treatment.
🏋️ 2× weekly resistance loading — additive to C10's anabolic signalsTrack Body Composition — Not Just Scale Weight
The scale does not distinguish fat loss from muscle loss. Two people who both lose 12 kg on semaglutide can have radically different outcomes depending on lean mass preservation: one loses 9 kg fat + 3 kg muscle; the other loses 12 kg fat + 0 kg muscle. Standard clinical monitoring of GLP-1 users tracks weight and metabolic labs — not body composition. If you have access to DEXA scan, InBody bioimpedance, or even basic anthropometric measures (waist circumference, mid-arm circumference), establishing a baseline before starting GLP-1 therapy and measuring every 12 weeks gives you the data to intervene if lean mass is eroding faster than expected.
📏 Baseline DEXA or bioimpedance before starting — recheck every 12 weeksHave the Body Composition Conversation With Your Prescriber
Most GLP-1 prescribers are tracking weight, HbA1c, blood pressure, and lipids. Fewer are actively monitoring lean mass. Raising the body composition question directly — "how are we going to track and preserve my muscle mass during this course?" — opens the conversation for muscle-preservation adjuncts including protein targets, resistance exercise referral, and potential discussion of supplements like C10. Physicians who prescribe GLP-1 drugs extensively are increasingly aware of the lean mass issue; what they often lack is a patient who has done the research to ask the right question.
🏥 Ask your prescriber: how are we tracking lean mass?The Evidence Base — Key Studies in Context
STEP 1 Trial — Semaglutide 2.4mg, 68-Week Outcomes Including Body Composition
The primary Phase III registration trial for semaglutide 2.4mg (Wegovy) in 1,961 adults with obesity or overweight plus comorbidities. Produced 14.9% mean body weight reduction vs. 2.4% for placebo. Body composition sub-study data confirmed that approximately 39% of weight loss was lean mass — establishing the clinical magnitude of the lean mass problem at GLP-1 drug scale.
Read the Study → NEJM, 2021 — Wilding JPH et al.SURMOUNT-1 Trial — Tirzepatide Body Composition Analysis
The Phase III registration trial for tirzepatide (Zepbound) demonstrating 20.9% mean weight reduction at 72 weeks with 5mg, 10mg, and 15mg doses. Body composition analysis confirmed meaningful lean mass reduction across dose groups, with the pattern broadly consistent with STEP 1 data: superior fat mass reduction, but substantial lean mass sacrifice as a proportion of total weight lost.
Read the Study → NEJM, 2022 — Jastreboff AM et al.MCTs Increase Muscle Mass and Function in Frail Older Adults — Ezaki & Abe, 2023
The combined data analysis of three randomized controlled trials confirming that 6g/day of C8/C10 MCTs over 90 days produced significant increases in arm muscle area (+1.4 cm²), grip strength (+1.6 kg), walking speed, knee extension, and body weight — while reducing fat mass — in adults with a mean age of 85. The directional outcome is the reverse of GLP-1-related body composition changes: lean mass up, fat mass down, functional capacity improved.
Read the Study → Frontiers in Nutrition, 2023Extracellular Signals Behind MCT Muscle Effects — Ezaki, 2024
This mechanistic follow-up published in Nutrients specifically examines the three extracellular pathways — SNS/PGC-1α mitochondrial biogenesis, acyl-ghrelin/GH secretion, and ketone nitrogen-sparing — that explain the 2023 clinical findings. The acyl-ghrelin pathway is particularly relevant to GLP-1 users, as GLP-1 receptor agonism suppresses ghrelin, and C10's documented ability to stimulate acyl-ghrelin may partially counteract GH-axis suppression caused by the drug.
Read the Study → Nutrients, 2024 — Ezaki O.C10 PPARγ Activation and Mitochondrial Biogenesis — Hughes et al., 2014
Cell-line study demonstrating that C10 — specifically, not C8 — activates PPARγ and increases mitochondrial citrate synthase and Complex I activity. PPARγ activation is upstream of PGC-1α, the master regulator of mitochondrial biogenesis. This establishes that C10's muscle-supporting effects operate at the genetic and organelle level — not simply as a caloric fuel — and represent a genuinely different intervention level than the appetite suppression mechanism of GLP-1 drugs.
Read the Study → Journal of Neurochemistry, 2014Lean Mass Regain After GLP-1 Discontinuation — STEP 1 Extension
The 20-week post-treatment follow-up of STEP 1 participants who discontinued semaglutide documented significant weight regain, with regained mass predominantly fat rather than lean tissue — confirming the ratchet mechanism of progressive body composition deterioration with GLP-1 cycling. This study establishes that the lean mass problem is not resolved by eventual drug discontinuation and underscores the importance of proactive lean mass preservation during the treatment period.
Read the Study → Diabetes, Obesity and Metabolism, 2022Protein Requirements During Caloric Restriction in Older Adults
A systematic review and meta-analysis establishing that protein requirements for muscle mass preservation during caloric restriction in adults over 60 are substantially higher than the standard RDA of 0.8g/kg/day — with current evidence supporting 1.2–1.6g/kg/day as the target range. Given that GLP-1 drugs suppress appetite and total food intake, meeting this target requires deliberate planning. C10's nitrogen-sparing mechanism reduces the minimum protein intake needed to maintain positive muscle protein balance by providing an alternative gluconeogenic substrate.
Reference: Current Opinion in Clinical Nutrition, 2015 — Deer & Volpi-
1
STEP 1 Trial — Semaglutide 2.4mg, 68-Week Body Weight and Composition — Wilding JPH, et al. "Once-Weekly Semaglutide in Adults with Overweight or Obesity." New England Journal of Medicine. 2021;384(11):989–1002.
DOI: https://doi.org/10.1056/NEJMoa2032183 -
2
SURMOUNT-1 Trial — Tirzepatide, 72-Week Body Composition — Jastreboff AM, et al. "Tirzepatide Once Weekly for the Treatment of Obesity." New England Journal of Medicine. 2022;387(3):205–216.
DOI: https://doi.org/10.1056/NEJMoa2206038 -
3
Weight Regain After Semaglutide Discontinuation — Lean Mass Ratchet — Wilding JPH, et al. "Weight regain and cardiometabolic effects after withdrawal of semaglutide." Diabetes, Obesity and Metabolism. 2022;24(8):1553–1564.
DOI: https://doi.org/10.1002/dom.14725 -
4
MCTs Increase Muscle Mass & Function in Frail Older Adults — Combined Analysis — Ezaki O, Abe S. "Medium-chain triglycerides (8:0 and 10:0) increase muscle mass and function in frail older adults: a combined data analysis of clinical trials." Frontiers in Nutrition. 2023;10:1284497.
DOI: https://doi.org/10.3389/fnut.2023.1284497 -
5
Extracellular Signals Behind MCT Muscle Effects — Mechanism Review — Ezaki O. "Possible Extracellular Signals to Ameliorate Sarcopenia in Response to Medium-Chain Triglycerides (8:0 and 10:0) in Frail Older Adults." Nutrients. 2024;16(16):2606.
DOI: https://doi.org/10.3390/nu16162606 -
6
C10 PPARγ Activation and Mitochondrial Biogenesis — Hughes SD, Kanabus M, Anderson G, et al. "The ketogenic diet component decanoic acid increases mitochondrial citrate synthase and complex I activity in neuronal cells." Journal of Neurochemistry. 2014;129(3):426–433.
DOI: https://doi.org/10.1111/jnc.12646 -
7
Protein Requirements in Older Adults During Caloric Restriction — Deer RR, Volpi E. "Protein intake and muscle function in older adults." Current Opinion in Clinical Nutrition & Metabolic Care. 2015;18(3):248–253.
DOI: https://doi.org/10.1097/MCO.0000000000000162

