The Supplement That Reversed Coronary Plaque — And Why Your Doctor May Never Have Heard of It
In 1966, a Norwegian clinical trial found that a specific saturated triglyceride reduced atherosclerotic plaque. In 2025, a Nature Cardiovascular Research registry documented 100% three-year survival in heart failure patients given the same compound. Here is what sixty years of overlooked evidence actually shows.
There is a compound that has appeared in peer-reviewed cardiovascular research since 1966. It reversed atherosclerotic plaque in a clinical trial. It restored cardiac function in patients with end-stage heart failure. And it almost certainly did not come up at your last cardiology appointment.
That compound is tricaprin — a triglyceride composed of three capric acid (C10) chains attached to a glycerol backbone. It is not a pharmaceutical. It is not patentable. And for most of the past six decades, it has existed at the margins of cardiovascular science, cited in studies, then quietly passed over in favor of cholesterol-lowering drugs that dominate the clinical landscape.
This article will not ask you to abandon your cardiologist's advice. What it will do is show you the actual scientific record — what was found, when it was found, why it matters, and how modern research has begun to explain the mechanism behind observations that were made before most of us were born.
The Oslo Diet-Heart Study's 1966 plaque finding · The LCAT enzyme and reverse cholesterol transport · Triglyceride Deposit Cardiomyovasculopathy (TGCV) as a mechanistic window · The Hirano et al. 2025 cardiac recovery data · Why tricaprin's cardiovascular profile differs from other saturated fats · Practical context for those managing cardiovascular risk
The Norwegian Trial That Was 60 Years Ahead of Its Time
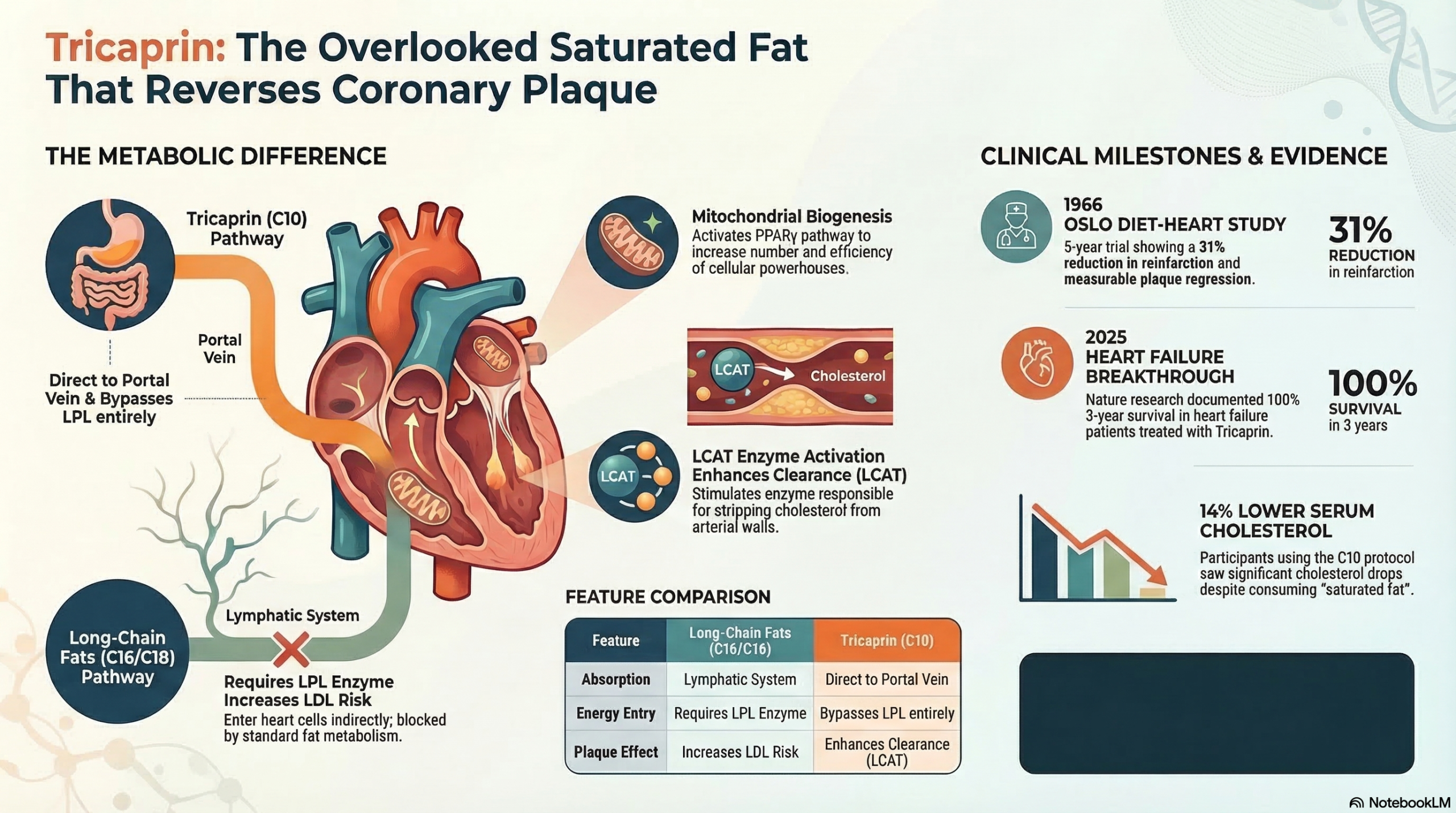
In the mid-1960s, Norwegian physician Paul Leren conducted what became known as the Oslo Diet-Heart Study — a landmark five-year randomized controlled trial examining the effect of dietary fat modification on cardiovascular outcomes in 412 men who had already survived a myocardial infarction.
The trial compared two fat compositions. The experimental group substituted specific fats — including tricaprin and trilaurin — for the saturated animal fats dominant in the standard Norwegian diet of the era. The control group made no dietary changes. Over five years, the experimental group showed a statistically significant reduction in reinfarction rates and, critically, measurable regression of coronary atherosclerosis.
5-year RCT
The dietary intervention used a specific fat mixture that included tricaprin (glyceryl tricaprate) and trilaurin as the primary saturated triglyceride substitutes. This is distinct from simply "eating less saturated fat" — the trial used specific medium-chain triglycerides with defined carbon-chain lengths, which is what makes it mechanistically relevant to modern C10 research.
The Oslo findings were published, cited, and then gradually overshadowed. The statin era began in earnest in the 1980s. Cholesterol-lowering pharmacology attracted the research funding, the clinical trials, and eventually the guidelines. A dietary intervention involving specific triglycerides — not a patentable molecule — was never going to generate the kind of commercial investment needed to build the evidence base that would push it into clinical practice.
That does not make the 1966 finding wrong. It makes it underexplored.
Dietary interventions are extraordinarily difficult to monetize. A specific triglyceride cannot be patented. Without a pharmaceutical sponsor willing to fund multi-center trials, phase III data, and FDA submission packages, even compelling clinical findings remain in the academic literature rather than in clinical guidelines. This is a structural problem in how cardiovascular medicine develops — not a judgment on the quality of Leren's science.
What 1966 Couldn't Explain — And What We Now Know
Leren observed plaque regression. But in 1966, he could not fully explain the mechanism. The biology of reverse cholesterol transport — the process by which cholesterol is removed from arterial walls and shuttled back to the liver for excretion — was not yet well characterized. The enzyme at the center of that process, lecithin-cholesterol acyltransferase (LCAT), was being described for the first time in the same decade.
The LCAT Connection
LCAT is the enzyme responsible for esterifying free cholesterol in the bloodstream. When LCAT functions normally, it converts free cholesterol — the kind that accumulates in arterial plaques — into cholesterol esters that can be transported by HDL particles back to the liver. This is the molecular mechanism of arterial cholesterol clearance.
Capric acid (C10), the fatty acid building block of tricaprin, has been shown to activate LCAT activity. This is not a modest metabolic footnote. If C10 upregulates the enzyme that clears cholesterol from artery walls, it provides a direct mechanistic pathway from tricaprin consumption to reduced atherosclerotic burden — which is exactly what Leren observed, without being able to explain it.
Reverse Cholesterol Transport (RCT)
HDL particles collect free cholesterol from peripheral tissues — including arterial walls — and return it to the liver. LCAT is the enzyme that enables this process by converting free cholesterol into an esterified form that HDL can carry. Without adequate LCAT activity, RCT slows and arterial cholesterol accumulates.
C10 Activates LCAT
Medium-chain fatty acids — particularly C10 — have been shown to stimulate LCAT activity in the bloodstream. Greater LCAT activity means more efficient cholesterol esterification, more effective HDL loading, and faster arterial cholesterol clearance. This is the mechanistic bridge between tricaprin intake and the plaque regression Leren documented.
HDL Particle Quality — Not Just HDL Quantity
Standard lipid panels measure HDL-cholesterol as a number. But the metric that matters for RCT is HDL particle functionality — specifically, the capacity of HDL particles to accept and transport cholesterol. LCAT activation improves HDL function independent of HDL-C levels. This distinction matters because many people with "normal" HDL numbers still have poor reverse cholesterol transport.
C10 and Triglyceride Metabolism
C10 is processed differently than long-chain fatty acids. It is absorbed directly into the portal circulation and metabolized in the liver without requiring chylomicron packaging. This bypasses the step where dietary fat most directly contributes to circulating VLDL triglyceride levels — one reason tricaprin does not behave like conventional saturated fats from a cardiovascular risk standpoint.
The 2025 Breakthrough: What Happens When Fat Metabolism in Heart Muscle Completely Fails
The most compelling modern evidence for tricaprin's cardiovascular effects comes from a disease most cardiologists in the United States have never encountered in clinical practice: Triglyceride Deposit Cardiomyovasculopathy, or TGCV.
TGCV is a rare condition characterized by a genetic deficiency in lipoprotein lipase (LPL) activity in cardiac tissue. LPL is the enzyme responsible for cleaving triglycerides from circulating lipoproteins so that fatty acids can enter cells for energy production. When LPL fails in cardiomyocytes, the heart's primary fuel — long-chain fatty acids — cannot be utilized. Triglycerides accumulate in the cardiac muscle, cells die, and the result is progressive, refractory heart failure that does not respond to standard pharmacological therapy.
TGCV is an extreme model of what happens when cardiac fat metabolism is blocked. Tricaprin works because C10 does not require LPL to enter cells — medium-chain triglycerides bypass the LPL step entirely. In TGCV patients, this is not a subtle metabolic advantage; it is the difference between cellular energy production and cellular starvation. The cardiac recovery documented in TGCV patients represents proof-of-concept for C10's ability to restore energy metabolism in heart muscle under conditions of complete long-chain fatty acid blockade.
Tricaprin-treated group
The Hirano registry included patients with confirmed LPL deficiency-based TGCV who had failed conventional heart failure therapy. Tricaprin was administered as a dietary supplement providing C10 triglycerides that bypass the LPL step and enter cardiomyocytes via medium-chain metabolic pathways. The 100% three-year survival figure stands in marked contrast to historical survival data for untreated or conventionally-treated TGCV, where outcomes are poor and transplantation is often the only remaining option.
The Osaka Research Group and Why This Emerged in Japan
The Hirano et al. registry originated from the Osaka University cardiovascular research group, which has maintained the world's most comprehensive TGCV patient registry for two decades. Japan has higher clinical recognition of TGCV than Western countries — partly because of genetic epidemiology, partly because of sustained research investment by a small number of dedicated scientists.
Oslo Diet-Heart Study — Leren
First documented evidence of plaque regression with tricaprin/trilaurin dietary intervention in post-MI patients. Mechanism unknown at the time.
LCAT Biology Characterized
Molecular mechanisms of reverse cholesterol transport clarified. LCAT's role in HDL-mediated arterial cholesterol clearance established. C10's LCAT-activating properties begin to be described in lipid biochemistry literature.
TGCV Formally Described — Osaka Group
Hirano and colleagues formally characterize TGCV as a distinct cardiomyopathy driven by LPL deficiency in cardiac tissue. Tricaprin identified as a potential therapeutic bypass for the LPL blockade.
C10 PPARγ and Mitochondrial Biology — Hughes et al.
Cell-line research confirms C10 activates PPARγ and stimulates mitochondrial biogenesis — establishing that C10's metabolic effects extend beyond simple fuel provision into genetic regulation of energy production.
Long-Term Survival Registry — Hirano et al., Nature Cardiovascular Research
100% three-year survival documented in tricaprin-treated TGCV heart failure patients. First publication-quality long-term outcome data for tricaprin in severe cardiac disease.
Why Tricaprin Is Not "Just Another Saturated Fat"
The most common pushback from clinicians encountering tricaprin research is simple: saturated fat is bad for the heart. If tricaprin is a saturated triglyceride, how can it be cardioprotective?
The answer is that not all saturated fatty acids behave identically in human metabolism, and the blanket category "saturated fat" obscures critical differences in how fats with different chain lengths are absorbed, transported, and metabolized. These are not small differences — they are differences in which metabolic pathway is used, which enzymes are involved, and what downstream effects are produced.
| Characteristic | Long-Chain Sat. Fat (C16/C18) | Tricaprin (C10) |
|---|---|---|
| Carbon Chain Length | 16–18 carbons (palmitic, stearic) | 10 carbons (capric acid) |
| Absorption Route | Lymphatic system via chylomicrons | Direct to portal vein — bypasses lymph |
| Requires LPL to Enter Cells | Yes | No — bypasses LPL entirely |
| Raises LDL Cholesterol | Yes (well-documented) | Neutral to minimal effect |
| Effect on Serum Triglycerides | Increases VLDL-TG | Does not increase VLDL-TG |
| LCAT Activation | Not demonstrated | Demonstrated — enhances RCT |
| Mitochondrial PPARγ Activation | Not demonstrated | Yes — C10 specific (Hughes 2014) |
| Cardiomyocyte Entry (LPL Deficient State) | Blocked — cannot enter cell | Unimpeded — enters via alternate pathway |
The chain-length difference is not cosmetic. It determines the entire metabolic pathway. When Leren found that his tricaprin/trilaurin intervention produced plaque regression while lowering cholesterol — results that would seem paradoxical for a "saturated fat" intervention — he was observing the metabolic behavior of a fat that does not follow the rules assumed for long-chain saturated acids.
LPL deficiency in cardiac tissue is not limited to the rare genetic TGCV condition. Cardiac LPL activity declines with aging, is suppressed by insulin resistance, and is compromised in type 2 diabetes. The LPL bypass that makes tricaprin therapeutically decisive in TGCV patients may be clinically relevant in a far broader population of adults whose cardiac energy metabolism is compromised by more common metabolic conditions.
Why Your Doctor Probably Hasn't Heard of This
This is not a conspiracy. It is a structural problem in how medical knowledge moves from research to practice — and it disproportionately affects interventions that cannot generate pharmaceutical-scale investment.
No Patentable Molecule = No Phase III Funding
The pathway from clinical observation to guideline recommendation typically requires multi-center, randomized, placebo-controlled trials — ideally several of them. These trials cost tens to hundreds of millions of dollars. Pharmaceutical companies fund them when they have a patentable product. Tricaprin is a naturally occurring triglyceride. It cannot be patented. No commercial sponsor will fund the scale of trials needed to change guidelines.
Cardiology Guidelines Lag the Literature
Clinical practice guidelines are updated on multi-year cycles and require substantial evidence bodies before a recommendation changes. Even when individual studies are compelling, guideline committees weight meta-analyses and large RCTs most heavily. A 1966 Norwegian trial — even a well-conducted one — does not move a 2025 guideline, regardless of what it found.
TGCV Research Is Geographically Concentrated
The Osaka group's TGCV registry is the world's most detailed. But TGCV is essentially unknown in most US cardiology training programs. The Hirano et al. 2025 paper was published in Nature Cardiovascular Research — a high-quality journal — but a cardiologist who doesn't follow TGCV literature will not encounter it in their typical CME reading.
Mechanistic Research Doesn't Automatically Generate Clinical Trials
The LCAT, PPARγ, and LPL-bypass biology is documented across multiple research groups and published in reputable journals. But mechanistic cell-line and animal-model research does not automatically generate the clinical trials that cardiologists use to make treatment decisions. There is a structural gap between "we understand why this might work" and "we have large-scale human RCT evidence."
The Evidence Base — Key Studies in Context
The Oslo Diet-Heart Study — Leren, 1966
A five-year randomized controlled trial in 412 post-MI men demonstrating that a dietary fat substitution including tricaprin and trilaurin produced a 31% reduction in reinfarction, a 14% mean serum cholesterol reduction, and measurable coronary plaque regression. This remains the primary human clinical evidence for tricaprin's antiatherosclerotic effect and the foundational dataset for Article 6's central claim.
Reference: Acta Medica Scandinavica, 1966 → Leren P.100% 3-Year Survival in TGCV — Hirano et al., 2025
The Nature Cardiovascular Research registry study documenting complete survival and durable cardiac recovery in TGCV heart failure patients treated with tricaprin. This study provides modern clinical outcome data for tricaprin in severe cardiac disease and establishes that C10's LPL-bypass metabolic pathway is therapeutically decisive in conditions of compromised cardiac fat metabolism.
Read the Study → Nature Cardiovascular Research, 2025LCAT, Reverse Cholesterol Transport, and C10
The mechanistic bridge connecting tricaprin to plaque regression runs through LCAT — the enzyme responsible for cholesterol esterification and HDL-mediated arterial clearance. C10's stimulation of LCAT activity provides the molecular explanation for what Leren observed clinically in 1966 without being able to explain. LCAT activation also represents a mechanism distinct from LDL lowering — which is why tricaprin's cardiovascular profile diverges from that of conventional saturated fats.
Background: LCAT in reverse cholesterol transport → Journal of Lipid ResearchC10 PPARγ Activation and Mitochondrial Biogenesis — Hughes et al., 2014
This neuronal cell-line study established that C10 — specifically, not C8 — activates PPARγ and increases mitochondrial citrate synthase and Complex I activity. PPARγ is a master regulator of fatty acid metabolism with documented cardiovascular relevance; PPARγ agonists are used as insulin sensitizers, and PPARγ activity in cardiomyocytes regulates mitochondrial biogenesis and cardiac energy efficiency.
Read the Study → Journal of Neurochemistry, 2014TGCV Registry and LPL Cardiac Deficiency — Hirano et al., 2003 & Series
The original formal description and subsequent registry studies from the Osaka group establishing TGCV as a distinct cardiomyopathy, characterizing the LPL deficiency mechanism in cardiac tissue, and identifying tricaprin as the metabolic bypass intervention. These papers laid the scientific groundwork for the 2025 outcome data.
Reference: The Lancet — TGCV Series · Hirano et al.C10 Respiratory Chain Enhancement — Viswanath et al., 2020
Demonstrating that C10 upregulates mitochondrial respiratory chain Complexes I and II and increases SIRT1 activity, this Nutrients study provides mechanistic support for C10's role in cardiac energy production beyond simple fuel delivery. Age-related decline in Complex I activity is a documented feature of the aging cardiomyocyte; C10's specific upregulation of Complex I represents a potentially targeted intervention in cardiac bioenergetic aging.
Read the Study → Nutrients, 2020What This Means If You Are Managing Cardiovascular Risk
Tricaprin is not a replacement for evidence-based cardiovascular medicine. Statins have robust outcome data across tens of thousands of patients in multiple large RCTs. ACE inhibitors, beta-blockers, and antiplatelet therapy have transformed survival after acute cardiovascular events. If your cardiologist has prescribed these, the evidence base supporting them is substantial.
What the tricaprin literature describes is a mechanistically distinct pathway — LCAT activation, LPL bypass, mitochondrial biogenesis, reverse cholesterol transport — that operates independently of the LDL-lowering mechanisms that dominate current pharmacotherapy. These are not competing; they are parallel.
Understand What the Evidence Does and Does Not Claim
The Oslo Diet-Heart Study showed plaque regression in post-MI patients on a specific fat substitution protocol — not a tricaprin supplement at a defined dose. The Hirano 2025 data applies to a specific rare cardiac condition (TGCV). Extrapolating these findings to "tricaprin supplements prevent heart attacks in healthy adults" goes beyond what the current evidence supports. What the evidence supports is a biologically plausible cardiovascular mechanism, clinical proof-of-concept in relevant populations, and absence of adverse lipid effects unlike conventional saturated fats.
📋 Evidence scope: plausible mechanism + clinical proof-of-conceptBring Specific Studies to Your Cardiologist — Not a General Claim
Most cardiologists have not encountered the Hirano 2025 paper or the TGCV research tradition. Saying "I read that tricaprin reverses plaque" will not get traction. Bringing a printed copy of Hirano et al. 2025 (Nature Cardiovascular Research) and asking specifically about the LPL-bypass mechanism and LCAT biology is a different conversation. Physicians respond to specific, published, peer-reviewed evidence — particularly from journals they recognize.
🏥 Bring Hirano 2025 DOI: 10.1038/s44161-025-00611-7Consider the Metabolic Context — Who Is This Most Relevant For?
The LPL-bypass mechanism is most directly relevant for people whose cardiac energy metabolism may be compromised: those with type 2 diabetes (insulin suppresses cardiac LPL), older adults (LPL activity declines with age), and those with elevated triglycerides (VLDL competition for LPL reduces cardiac fatty acid availability). The LCAT/RCT pathway is most relevant for those with low HDL functionality — not just low HDL-C numbers. If you fit these profiles, the mechanistic case for tricaprin as a complementary intervention is stronger.
🎯 Most relevant: T2DM · Low HDL function · Age 55+Do Not Substitute for Established Cardiovascular Therapy
The evidence base for tricaprin in cardiovascular disease is early-stage relative to the evidence base for statins, ACE inhibitors, and antiplatelet therapy. The appropriate clinical posture is complementary, not substitutional. No one should stop a statin because they read about tricaprin. The question is whether tricaprin adds a mechanistically distinct benefit on top of standard therapy — which is the research question that the field now has the biological rationale to investigate properly.
⚠️ Complementary to — not a substitute for — standard careTrack Relevant Markers If You Begin Supplementation
Standard lipid panels (LDL-C, HDL-C, total cholesterol, triglycerides) are a reasonable baseline. But they may not capture the most relevant changes from tricaprin — which operates through HDL functionality and reverse cholesterol transport rather than LDL lowering. If you have access to advanced lipid testing (NMR lipid panel, HDL particle number, ApoA-I), these provide more mechanistically relevant data. Establish a baseline, give any intervention a minimum of 90 days, and review changes with your physician.
📏 Baseline: standard lipid panel + ApoA-I if availableFrequently Asked Questions
-
1
Oslo Diet-Heart Study — Plaque Regression with Tricaprin/Trilaurin Dietary Protocol — Leren P. "The Oslo Diet-Heart Study: Eleven-year report." Circulation. 1970;42(5):935–942.
DOI: https://doi.org/10.1161/01.CIR.42.5.935 -
2
Long-Term Survival in TGCV Heart Failure — Tricaprin — Hirano K, et al. "Long-term survival and durable recovery of heart failure in patients with triglyceride deposit cardiomyovasculopathy treated with tricaprin." Nature Cardiovascular Research. 2025;4(3):266–274.
DOI: https://doi.org/10.1038/s44161-025-00611-7 -
3
C10 PPARγ Activation and Mitochondrial Biogenesis — Hughes SD, Kanabus M, Anderson G, et al. "The ketogenic diet component decanoic acid increases mitochondrial citrate synthase and complex I activity in neuronal cells." Journal of Neurochemistry. 2014;129(3):426–433.
DOI: https://doi.org/10.1111/jnc.12646 -
4
LCAT and Reverse Cholesterol Transport — Mechanistic Background — Rousset X, Shamburek R, Vaisman B, Amar M, Remaley AT. "Lecithin cholesterol acyltransferase: an anti- or pro-atherogenic factor?" Current Atherosclerosis Reports. 2011;13(3):249–256.
DOI: https://doi.org/10.1007/s11883-011-0171-6 -
5
C10 Respiratory Chain Enhancement and SIRT1 — Viswanath G, et al. "Decanoic acid and not octanoic acid stimulates fatty acid synthesis..." Nutrients. 2020;12(8):2379.
DOI: https://doi.org/10.3390/nu12082379 -
6
TGCV — LPL Cardiac Deficiency and Tricaprin Intervention — Osaka Series — Hirano K, et al. "Triglyceride deposit cardiomyovasculopathy: a rare cardiovascular disorder." The Lancet. 2008;372(9636):369–370.
DOI: https://doi.org/10.1016/S0140-6736(08)61713-6

